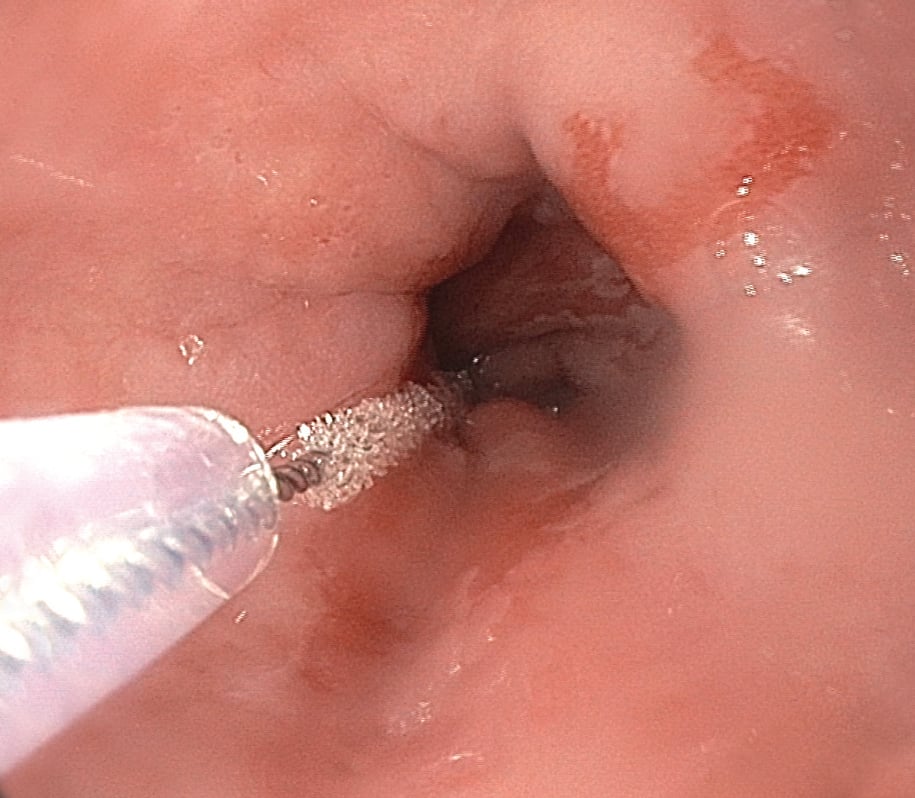
For greater inter-observer agreement than standard histopathology on forceps biopsy samples.
With WATS3D, areas identified by the neural network as the most likely to be dysplastic, pathologists are able to quickly identify and grade dysplasia in higher percentage of positive cases and with far greater diagnostic accuracy.
Traditional esophageal histopathology on forceps biopsy specimens are well-known to suffer from high interobserver variability and high miss rates of dysplasia and cancer.
WATS3D pathologists gathers information from both the 3D images as well as the cytology specimens using histocytology techniques to make more accurate diagnoses, and genetic testing allows pathologists to consistently make a diagnosis based on the most advanced science available.
Prashanth R. Vennalaganti, MD , Vijay Naag Kanakadandi, MD, Seth A. Gross,MD, Sravanthi Parasa, MD, MPH, Kenneth K. Wang, MD, Neil Gupta , MD, MPH and Prateek Sharma.
Am J Gastroenterol, 28 April 2015; doi: 10.1038/ajg.2015.116